Pharma's Curious Case of Missing Vaccine Efficacies in the COVID-Recovered.
In an era of limitless data and instant analysis, why are vaccine efficacies for the COVID-recovered omitted in pharma-sponsored studies? And, why aren't our leaders asking questions ?
Much has been said recently about vaccine “misinformation”. But, what can be said about tactical omission of true and critical information that goes against a high-stakes policy position? What if Pharma-sponsored vaccine studies shrewdly omit or suppress information that does not fit their desired narrative? What if our agencies are letting this pass, in order to preserve their inflexible policy positions?
Let us look at the specific case of vaccine efficacy (VE) in COVID-recovered persons. Both Pfizer and Moderna have sponsored studies that can clearly answer whether vaccination provides protection previously infected persons. However, each of these studies’ publications conveniently omit or downplay this data, while providing other less impactful observations. If these numbers justified vaccination of the COVID-recovered, what is there to hide?
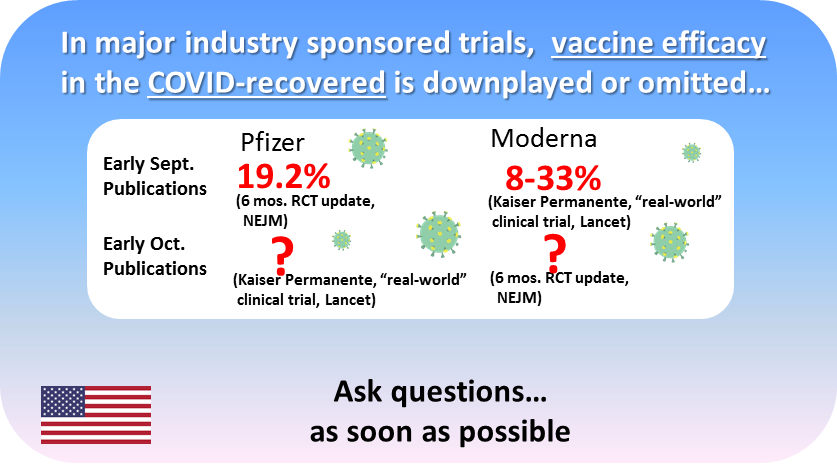
In their initial December 2020 FDA applications, both Pfizer and Moderna could NOT claim a statistically significant benefit to vaccination in the COVID-recovered. Both Pfizer and Moderna sponsored studies, released in early September — that demonstrated paltry VE in the COVID-recovered (19.2% and 8-33%, respectively). Though the numbers were published, these results were buried deep in large tables, and received minimal to no attention in the textual discussion by the authors. Later in September, subsequent Pfizer and Moderna sponsored studies were released — each that had the data to provide a significant update on VE in the COVID-recovered, but these metrics were entirely omitted in publication. These are not “small” studies, but are in fact, the extension of the “pivotal” trials used for FDA approval, and large “real-world” vaccine trials with millions of subjects.
This is a very big issue — because nearly 120 MILLION Americans likely have had symptomatic or asymptomatic COVID-19 infection — with a large fraction likely having durable natural immunity. The CDC also believes that a large fraction of this population are the remaining vaccine “hesitant”, and a barrier to their goal of “full vaccination”. Thus, the CDC has been persistent on their claim that previously infected persons should get vaccinated. The problem is that they have difficulty basing this on EVIDENCE, as I explain in a previous Substack article. So, the CDC embarks on commissioning their own studies so they can cite "evidence” to back their predetermined claims — hence, the now famous “Kentucky” and “Hospital” studies, which use convoluted and flawed methodologies to answer simple epidemiological questions.
Given that some other studies (the CDC’s “KY” study and the Israeli Gazit study) have shown approximately a 50% VE in the COVID-recovered, if Pharma-sponsored studies found anything better, why wouldn’t they publish it and socialize it to the public? Certainly, a strong COVID-recovered VE would bolster each companies claim that vaccination is required in the COVID-recovered. However, if the VE for the COVID-recovered are significantly lower (or even negative) — it would put into question the CDC’s inflexible position on vaccination of this subgroup. It would also add weight to the challenges on inflexible mandates that may not benefit everybody (such as the recent stay on the OSHA mandate, ordered 5th Circuit Court of Appeals). As such, these “incidental” omissions should raise serious questions of Pharma, and by our government agencies and officials.
As a physician citizen, I feel it is one of my greatest civic duties to ask these difficult questions of our agencies, and collaborating industries. Maybe these numbers will ultimately show favorable VE for the COVID-recovered, and support the CDC’s otherwise flailing position. Honestly, I hope that it does — as, it would finally scientifically justify an inflexible policy, and mitigate our worst fears of political influence. However, the less than forthcoming approach of the CDC and Pharma companies raises more questions than it answers. So, let us look at the Pfizer and Moderna cases separately…
The Pfizer Case
In the original VRBPAC briefing (December 10th, 2020) for the COVID-19 vaccine, Pfizer presented its initial clinical trial of ~40,000 patients (20,000 in each arm), demonstrating an overall efficacy of 94.6%. However, with regards to the COVID-recovered, they could not be so certain, and noted the following (pg.28):
From this paragraph, we can glean that there were actually MORE infections in the vaccination group (10) compared to the placebo group (9), but only after applying the definition of infection after 7 days of the second dose (21 days after the first dose), could they arrive at 9 infections in the placebo group and only 1. Nonetheless, in the “data not shown” from above, they arrive at a -7.1% vaccine efficacy, because the same number of infections (1) occurred in a smaller surveillance time. However, because of the low case count, this was statistically “insignificant” (pg 27) :
Still, in their written narrative, they manage to make a convoluted case for vaccination in the recovered, stating “While limited, these data do suggest that previously infected individuals can be at risk of COVID-19 (i.e. reinfection) and could benefit from vaccination.” In other words, vaccination was supported not because the vaccine is effective in this group, but only because the possibility for reinfection exists. This is a subtle logical mind trick, attempting to deflect attention from the weak efficacy. Nonetheless, because it was early in the process, we all gave Pfizer the benefit of the doubt.
However, on September 16th 2021, Pfizer released an update to this study in the prestigious NEJM, to report on the safety and efficacy of the vaccine after 6 months. Overall, they found an overall VE of 91.3%. However, in the main text of the article, they skirted the issue of VE in the COVID-recovered. In fact, they explicitly state that “previous infection conferred approximately a 72.6% protection” but specifically OMIT the discussion of the vaccine efficacy in the COVID-recovered:
In other words, they downplay the ineffectiveness of the vaccine (19.2%), and instead tactically imply the relative weakness of natural immunity (72.6% vs. 91.3%) In fact, you have to dig down into Table S5, attached to the Supplementary Index, on page 12 of 18, to find the COVID-recovered VE:
The VE for all COVID-recovered persons was a ridiculously low 19.2%, and still statistically insignificant (in fact, there is a higher probability that the VE is negative rather than positive!). The question is why would this key fact would NOT be discussed in the main text during discussion of vaccination in the previously infected? In fact, it seems they go out of their way to avoid disclosure of this data. While all other subgroups (age, race, ethnicity, country, gender) demonstrated adequate efficacy, the COVID-recovered was the only major subgroup that indicated suboptimal efficacy! To the cynic, this could be construed as a passive attempt to divert attention from an embarrassing result. Notably, this suboptimal finding has not been publicized.
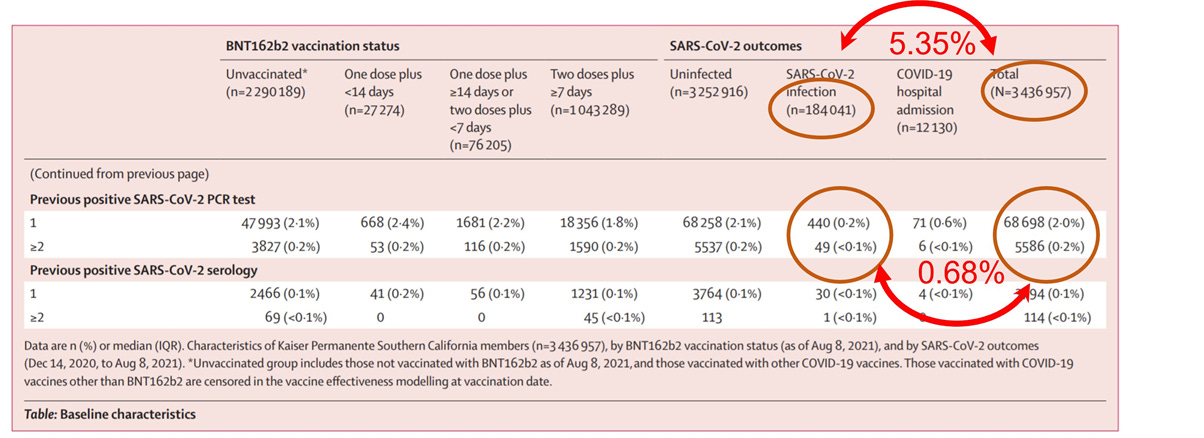
In early October, authors released a pre-print (the paper is behind a firewall) of another Pfizer-sponsored “real-life” study set in the Kaiser Permanente system, of more than 3.4 million individuals, with roughly 73,000 previously infected persons — a very large subgroup. In their Table 1 reporting baseline characteristics, they describe the previously infected subgroup, demonstrating a significantly lower rate of reinfection (0.68%) compared to the infection rate of the overall population (5.35%), despite similar vaccination proportions (72% vs 66%). This observation on the effectiveness of natural immunity, however, is not reported in the main text — but can only be calculated from the raw data.
However, the granular data required to calculate VE for the COVID-recovered are simply not given, though obviously this data was collected. To be fair to this author group, a lot of the data for the other subgroups are not given as well (e.g. comorbidities, gender, race), so it may just be a coincidence. But at the very least, this author group (and the sponsor, Pfizer) holds vitally important data to 40-120 million Americans, and should feel compelled to produce more specific VEs, and particularly for the COVID-recovered. Its omission, in the setting of being a Pfizer funded study, is at least, curious.
Would anybody believe that, if the preliminary analysis demonstrated significantly positive VE in the COVID-recovered, that it would not be enthusiastically published and socialized?
So, for Pfizer, long-term VEs for the COVID-recovered have only been published in one study (19.2%), with large missed opportunities to present this data with the real-world study. A COVID-recovered VE this low would not have met approval threshold , as published a priori by the FDA, which would require a 50% estimate (with a lower confidence bound of 30%):
Our government agencies and government representatives should be seriously asking the question as to why Pfizer’s sponsored real-life study would omit a critical number to millions of previously-infected Americans, and how a best estimate of a 19.2% VE is sufficient to justify mandated vaccination in this group.
As a separate issue, this was never brought up by the FDA VRBPAC in their discussion of the Pfizer vaccination in 5-11 year olds, of whom nearly 38% may be COVID-recovered. In addition to the already low rates of infection and morbidity in this age-group, COVID-recovered children likely have even lower risk of infection, combined with weak vaccination efficacies, to likely provide minimal absolute benefit (as per my previous article).
Who is asking these questions?
The Moderna Case
Similarly, Moderna presented their EUA case to the VRBPAC on December 17, 2020. In their study of about 30,000 patients (15,000 in each arm), only 341 and 334 previously infected persons were included in vaccine and placebo groups, respectively.
Because the rate of reinfection was so low, and the “prior infection” groups were so small, there was only 1 reinfection in the placebo group, and none in the vaccination group, leading to to a 100% VE — albeit, statistically meaningless. The authors of the EUA VRBPAC briefing conclude:
Certainly, this is a reasonable conclusion. But notably, in this first presentation, there is no hesitation to display this result, even if it is statistically insignificant — well, because, it is not detrimental to Moderna’s narrative — after all, its 100% VE ! However, in late September 2021, Moderna sponsors an update to this specific study group, in an NEJM publication. The study was very positive, and found a 93.2% overall VE for symptomatic infection, after 6 months. This, of course, is excellent for COVID-naive individuals.
But, in this 6 month update, they EXCLUDE any report of vaccine efficacy in COVID-recovered persons. This omission is very suspicious for TWO reasons:
First, the authors include a VE for virtually every other subgroup regardless of size or significance— including VEs for “warehouse shipping and fulfillment” workers (N=158 and 174) and “border protection and military personnel” (N=66 and 64). They even include a VE for liver disease patients (N=90 and 100) and “Native Hawaiian and other Pacific Islanders” (N=31,36) which were statistically insignificant and incalculable (e.g. “NE”) — but they see it necessary to COMPLETELY OMIT the “prior infected”! (N=341, 334). Remember, they had no problems reporting the statistically insignificant “100% VE” in the VRBPAC briefing. This should raise significant regulatory eyebrows.
The paper does not omit the prior infected completely, however. Though deciding to omit the prior infected from the EFFICACY analysis, they provide deep focus on the prior infected in the Safety Analysis portion of the study (!), to conclude vaccination is “safe” in the previously infected :
So to review — when it was convenient to Moderna in the VRBPAC briefing, they had no issue publishing a statistically insignificant 100% VE in the prior infected . . . but, in the 6 month update, they omitted publishing the VE for the same (prior infected group), even though they analyzed VE for every other subgroup. They also presented extensive “safety” analysis and discussion for the previously infected — but still, neglect publishing the VE in the same group. These selective and rather acrobatic presentations/omissions should raise red flags!
But we are not completely blind to a possible VE in COVID-recovered persons vaccinated with Moderna. In fact, a slightly earlier “real-life” Moderna-funded study conducted in the Kaiser Permanente system (as was done with Pfizer, above), did in fact study 60,000 previously infected individuals, and found a VE of 8.2-33.6% (0-65.8%)- the variation here depended on definition of diagnosis history), while finding a VE in naive persons of 89.3%. In other words, VE for the COVID-recovered is significantly lower than expected.
(N.B. Another interesting point here is that the low-end confidence intervals for the VEs in the previously infected were truncated at 0.0,, when the the negative tail should probably extend into the negative range) !
Without question, these are suboptimal VEs for the previously recovered. Though this suboptimal VE is at least mentioned in the manuscript text , the authors state that:
“Our study suggests that among individuals with history of COVID-19, mRNA-1273 vaccination may provide additional benefit (8·2-33·6%) beyond protection from natural infection, supporting current guidance that individuals with history of COVID-19 should be vaccinated”
In other words, they want you to believe that any VE greater than 0% would justify vaccination in the previously infected. This concept, of course, is preposterous — again, to remind you, even the FDA’s pre-approval guidance, they required at least 50% efficacy for approval:
So, ultimately, the only real VE we have for Moderna vaccination in the previously infected is between 8-33%, and would have been subthreshold even by FDA standards. Where are our agencies and representatives probing this critical observation?
We Need to Ask the Difficult Questions
In order to make the scientifically logical argument for mandating vaccination in the previously infected, the authorities need to prove at least two necessary claims: 1) That prior infection does not confer sufficiently similar protection to vaccination against SARS-CoV-2 infection, and 2) that vaccination produces additional and meaningful benefit over intrinsic natural immunity. This latter question, of VE in the previously infected, is critical to this claim.
To date, the CDC does not have strong data that VE is sufficiently strong in the previously infected. In their “brief”, even though they claim…
“[substantial] epidemiological evidence indicate that vaccination after infection significantly enhances protection and further reduces risk of reinfection, which lays the foundation for CDC recommendations.”
…their cited evidence comes down to their own small, flawed “KY” study, the Israeli Gazit study (which actually also shows substantially superior natural immunity 6-27x), and a recent Indian study (which found a small and statistically insignificant difference to vaccination, and does not even study a vaccine available in the U.S.!). Further, the CDC “brief” OMITS these most germane Pfizer and Moderna studies, demonstrating embarrassingly low 19.2% and 18-33% VE, respectively. If the CDC were objective and intellectually honest, they would have quoted these otherwise respectable studies in their brief — as it precisely addresses the crucial question. However, the result does not fit their desired narrative — and so it was omitted. Thus, their recommendation has no credible “foundation”.
Subsequent to these initial “19.2"%” and “18-33%” publications, both Pfizer and Moderna have gone radio silent on VE in the COVID-recovered. WHY WOULD PFIZER AND MODERNA NOT PUBLISH PRIOR INFECTED VE IN SPONSORED STUDIES WHERE THEY CLEARLY AND EASILY COULD HAVE??? If these VE’s positively reflected on mRNA vaccine efficacy, it is almost certain they would have published these to put the issue to rest. However, the omission of these critical numbers has to at least raise suspicion that the long-term VE in the previously infected is more modest or weaker than expected. Is there another explanation?
After all, if the VE for the COVID-recovered is greater than 50%, Pharma and the CDC would only bolster their policy position, and may convince the remaining unvaccinated to get vaccinated. If the VE is somewhat lower, then it would reinforce the current literature and would not compel a change in their position. However, if the VE is very low or negative, then there is the very real possibility that these vaccines actually may increase the risk of infection and do harm in the COVID-recovered, a completely untenable, if not fraudulent circumstance. These VEs must be published to negate this diabolical possibility.
I personally reached out to the authors of these studies, from whom I appreciated their polite and unexpected replies. While the studies are pharma-funded, the investigators are well-intended research scientists at respectable institutions. One author suggested the results would not have met statistical significance, which has really never been an excuse for omission. Another author suggested the data is complex and subsequent analysis will be published at a later time. I do want to take fellow physicians and scientists at their word. However, with hundreds of millions of COVID-recovered adult and children worldwide — I would hope that the authors feel a compelling ethical obligation to release these results in a timely, if not immediate, fashion. And, with more than ~10M COVID-recovered 5-11 year olds now facing possible mandates, there is a crucial moral imperative to RELEASE THESE RESULTS.
Pfizer and Moderna have received significant government subsidies and liability waivers for the COVID-19 vaccines. While we do not expect these companies to serve outside of their corporate interests, OUR government agencies, such as the FDA and CDC, should be holding these companies to the highest levels of transparency. Unfortunately, the CDC does not appear to be an objective player, and seems wedded to political endpoints, rather than scientific transparency and objectivity.
Again, it is one of our greatest civic duties to ask difficult questions of our representatives and authorities, particularly in the arena of public health. I have reached out to the authors, wrote letters to editors, emailed CDC/FDA directors, and even reached out to key U.S. Senators on the subject — but have gotten no traction. I am hoping, by virtue of this article, that anybody who reads this that can help socialize and resolve this curiosity. I am simply asking for release of the COVID-recovered data in these two studies (Pfizer and Moderna), that can help definitively answer the question of vaccination in the COVID-recovered.
If the CDC and Pharma can show substantial vaccine efficacy in the COVID-recovered, they would only promote their goal of minimizing vaccine hesitancy, and encourage the remaining to get vaccinated. Pharma, the CDC, the Administration, and the American people would have everything to gain! However, the failure to report this data, however, only fuels the clandestine perception, and weakens our trust in the system. Release the COVID-recovered VE’s !
Thank you for reading!
DISCLAIMER: This article represents my opinion only, and not of any organization I am affiliated with. It based upon best efforts to compile and analyze the data and evidence. The intended use is for policy discussion purposes only. It is not a substitute for advice from a personal physician. Please consult your personal physician for health advice.














As a parent of a covid recovered child, I am especially concerned about the potential vaccine mandate for all children in public & private schools in NY (an assembly bill A08378 has been introduced) I have been following your substack closely and wondering if any studies of Covid recovered & vaccinated children are in progress and when might we have more information? I feel at this point in time, I can’t in good conscience allow my child to be vaccinated without any AE data.
Really well done! AS you say, this data should not be left out due to oversight. this category is HUGE. One might even argue central.